What is gaseous state ?
-
-
What is the gaseous state?
A gaseous state is understood as one of the four states of aggregation of matter, together with the solid, liquid and plasma states. Substances in the gaseous state are called “gases” and are characterized by having their constituent particles little joined together, that is, expanded along with the container where they are located, to cover as much as possible the available space.
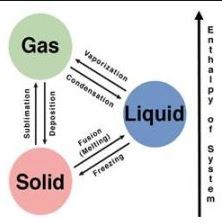
The latter is due to the fact that they have a very mild force of attraction, which means that the gases lack a defined shape and volume, taking those from the container that contains them, and also having a very low density, since their particles are in a state of relative disorder, moving or vibrating very quickly.
In this way, the molecules that make up the matter in a gaseous state do not manage to hold each other firmly and stay together with great difficulty, being even less affected by gravity, compared to solids and liquids: that allows them to float up. Despite its almost zero cohesion, the gases have an enormous capacity to be compressed, which is often carried out during their industrial treatment for transport.
The physical properties of a given gas (color, taste, smell) may vary depending on the elements that constitute it or that are dissolved in it. For example, ordinary air is colorless and odorless and tasteless, while hydrocarbon gases such as methane have a typical unpleasant smell and may have color.
-
-
-
- From liquid to gas: evaporation. This process occurs daily, just by injecting heat energy into the liquid until its temperature exceeds the boiling point (different depending on the nature of the liquid). Water, for example, boils at 100 ° C and passes into a gas (steam).
- From solid to gas: sublimation. In some cases, solids can go directly to the gaseous state without first going through liquidity. A perfect example of this occurs at the planet’s poles, where the temperature is so low that the formation of liquid water is impossible, but still, ice and snow sublimate directly to the atmosphere.
- From gas to liquid: condensation. This process is contrary to evaporation and has to do with the subtraction of heat energy from the gas, causing its particles to move more slowly and come together with greater force. It is what happens in the atmosphere when, when moving away from the earth’s surface, the water vapor loses temperature and forms clouds that, eventually, precipitate the drops of water back towards the ground: rain.
- From gas to solid: reverse sublimation. This process can also be called crystallization, in some specific contexts. And it takes place under specific pressure conditions, which force the particles of gas to gather beyond what they commonly are, bringing them directly to the solid-state without first going through liquidity. An example of this is the semi-solid frost, which appears on the windows of a winter day.
-
You May Also Like:
- What are Autotrophs?
- What are biogeochemical cycles?
- What is Nitrogen Cycle?
- What are Abiotic And Biotic Factors Of The Desert?
