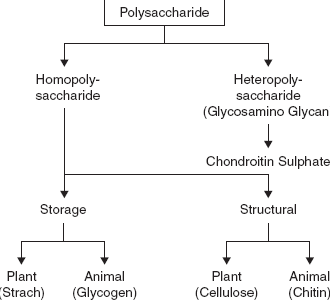
Types of Polysaccharides
The following lines highlight the three main types of Polysaccharides. The types are: 1. Food Storage Polysaccharides 2. Structural Polysaccharides 3. Mucosubstances
Type # 1. Food Storage Polysaccharides:
These are those polysaccharides which serve as reserve food. At the time of need, storage polysaccharides are hydrolysed. Sugars thus released become available to the living cells for production of energy and biosynthetic activity. The main two storage polysaccharides— starch and glycogen.
1. Starch:
It is a storage polysaccharide of most the plants.The human beings obtain it from cereal grains (e.g., rice, wheat), legumes (pea, gram, beans), potato, tapioca, banana etc. It is polyglucan homosaccharide and it is formed as an end product of photosynthesis. Starch is stored either inside chloroplasts or special leucoplasts called amyloplasts. Starch occurs in the form of microscopic granules called starch grains.
Starch grains may occur singly or in groups. The two types are known as simple and compound starch grains. The Starch grains may be rounded, oval, polygonal or rod shaped in outline (Fig. 9.6). Each grain has a number of shells or layers arranged in concentric or eccentric fashion around a proteinaceous point called hilum.
Starch consists of two components, amylose and amylopectin . Amylose is more soluble in water than amylopectin. In general, 20-30% of starch consists of amylose and the rest as amylopectin. Waxy starch of some varieties of Maize and other cereals consists entirely of amylopectin. On the other hand, the starch of some varieties of Pea having wrinkled surface may have as much as 98% of amylose.
Both amylose and amylopectin are formed by the condensation of α -D-glucose (pyranose forms). Amylose is in the form of a continuous straight but helically arranged chain where each turn contains about six glucose units.
The successive glucose units are linked together by 1-4 α-linkages, that is, the link is between carbon atom 1 of one and carbon atom 4 of the other (Fig. 9.8). A molecule of water is lost during the formation of the linkage. The straight chain of amylose consists of 200-1000 glucose units.
Amylopectin contains a large number of glucose units (2000-200,000). Besides a straight chain it bears several side chains which may be branched further.
Branching is usually at intervals of about 25 residues. At the place of origin of a side chain, the carbon atom 6 of a glucose residue of a straight chain is linked to the carbon atom 1 of the first glucose unit of side chain (1-6 α-link- age). Wolform and Thompson (1956) have also reported 1 → 3 linkages in case of amylopectin.
Test:
Amylose fraction gives blue-black colour with iodine solution (Iodine-Potassium iodide solution) while amylopectin fraction gives red-violet colour.
2. Glycogen:
It is a polysaccharide food reserve of animals, bacteria and fungi. Glycogen is popularly called animal starch. Glycogen is mainly stored inside liver (up to 0.1 kg) and muscles. In shape the complex carbohydrate appears as ellipsoid flattened granules that lie freely inside the cells. The polysaccharide gives reddish colour with iodine. Chemically, it is similar to starch.
It has about 30,000 glucose residues and a molecular weight of about 4.8 million. Glucose residues are arranged in a highly branched bush like chains. There are two types of linkages 1-4 α -linkages in the straight part and 1-6 linkages in the area of branching. The straight part of it is helically twisted with each turn having six glucose units. The distance between two branching points is 10-14 glucose residues.
3. Inulin:
It is a fructan storage polysaccharide of roots and tubers of Dahlia and related plants. Inulin is not metabolized in human body and is readily filtered through the kidney. It is, therefore, used in testing of kidney function, especially glomerular filtration.
Type # 2. Structural Polysaccharides:
They are polysaccharides that take part in forming the structural frame work of the cell walls in plants and skeleton of animals. Structural polysaccharides are of two main types: chitin and cellulose.
1. Chitin:
It is the second most abundant organic substance. Chitin is a complex carbohydrate of heteropolysaccharide type which is found as the structural component of fungal walls and exoskeleton of arthropods. In fungal walls, chitin is often known as fungus cellulose. Chitin is soft and leathery. Therefore, it provides both strength and elasticity. It becomes hard when impregnated with certain proteins and calcium carbonate.
In chitin, basic unit is not glucose but a nitrogen containing glucose derivative known as N-acetyl glucosamine. Chitin has an un-branched configuration. Monomers are joined together by 1- 4 β-linkages (Fig. 9.9). Adjacent residues lie at 180°. Molecules occur in parallel and are held together by hydrogen bonds.
2. Cellulose:
It is fibrous homopolysaccharide of high tensile strength which forms a structural element of cell wall in all plants, some fungi and protists. Tunicin of tunicates (=ascidians) is related to cellulose (also called animal cellulose).
In absolute terms, cellulose is the most abundant organic substance of the biosphere forming 50% of carbon found in plants. Cotton fibres have about 90% of cellulose while wood contains 25-50% cellulose. The other materials of the cell wall include lignin, hemicellulose, pectins, wax, etc.
Cellulose molecules have un-branched and linear chains unlike the branched and helical chains of starch and glycogen. A chain of cellulose molecule contains 6000 or more glucose residues.
The successive glucose residues are joined together by 1-4 β-linkages (Fig. 9.10). Consequently alternate glucose molecules lie at 180° to each other. Hydroxyl groups of glucose residues, therefore, project in all directions. The molecular weight of cellulose ranges between 0.5 to 2.5 millions.
Cellulose molecules do not occur singly. Instead a number of chains are arranged in close antiparallel fashion. The molecules are held together by intermolecular hydrogen bonds between hydroxyl group at position 6 of glucose residues of one molecule and glycosidic oxygen between two glucose residues of the adjacent molecule.
There is also intermolecular strengthening of the chain by the formation of hydrogen bonds between hydroxyl group at position three and oxygen atom of the next residue. About 2000 cellulose chains or molecules are packed together to form a micro fibril visible under the electron microscope.
Importance of cellulose
(1) Cellulose constitutes the bulk of human food. However, due to being polymer of β-glucose, cellulose is not acted upon by amylases present in human digestive juices. In humans, cellulose has a roughage value which keeps the digestive tract in functional fitness.
(2) Cellulose is an important constituent of diet for ruminants like cows and buffaloes. The stomach of ruminants contain micro-organisms capable of digesting or breaking down cellulose. Termites and snails also possess micro-organisms in their gut for this purpose.
(3) Microbes are used in producing soluble sugars from cellulose. The sugars are then allowed to undergo fermentation for obtaining ethanol, butanol, acetone, methane, etc.
(4) Cellulose rich wood is employed in building furniture, tools, sports articles, paper etc.
(5) Depending upon the percentage of cellulose present in the fibres, the latter are used in textiles (e.g., Cotton, Linen), preparation of sacs (e.g., Jute) or ropes (e.g., Hemp, China Jute, Deccan Hemp).
(6) Rayon and cellophane are formed of cellulose xanthate.
(7) Cellulose acetate is obtained by treating wood pulp with acetic acid, acetic anhydride and a catalyst. Cellulose acetates are used in preparing fibres for double knits, tericot, wrinkle proof, and moth proof clothing. Cigarette filters are also prepared from these fibres. Other uses of cellulose acetates include preparation of plastics and shatter proof glass.
(8) Cellulose nitrate is used in propellent explosives.
(9) Carboxymethyl cellulose is used as emulsifier and smoothening reagent of ice creams, cosmetics and medicines.
Type # 3. Mucosubstances:
Mucilage, mucus or slime forming substances are called mucosubstances. They are of two types, mucopolysaccharides and mucoproteins (= glycoproteins).
Mucopolysaccharides:
They are slimy substances or mucilages which possess acidic or aminated polysaccharides formed from galactose, mannose, sugar derivatives and uronic acids. Mucopolysaccharides or mucilages are quite common in both plants and animals.
They can be observed by cutting the unripe fruits of Okra (Lady’s finger, vern Bhindi) or soaking the husks or seeds of Plantago ovata (Plantain, vern. Isabgol). Mucopolysaccharides occur inside the plant cell walls, outside the cells or bodies of bacteria, blue-green algae and many aquatic plants, cementing layer between cells, inside body fluids, connective tissues and cartilages.
The important functions of mucopolysaccharides are as follows:
(1) They bind proteins in the cell walls and connective tissue.
(2) Water is held in the interstitial spaces due to mucopolysaccharides.
(3) Mucopolysaccharides occur in the cell walls of bacteria and blue-green algae. They form a layer of mucilage around them as well as other aquatic plants and protects the organisms against rotting effect of water, prevents desiccation, growth of epiphytes and attack of pathogens.
(4) They provide lubrication in ligaments and tendons.
(5) As keratan sulphate (acetyglucosamine + galactose + sulphuric acid) they occur inside skin and cornea providing both strength and flexibility.
(6) Chondroitin sulphate (glucuronic or iduronic acid + acetyl aminogalactose) is the mucopolysaccharide found in the matrix of cartilage and connective tissue for support and elasticity.
(7) Hyaluronic acid (glucuronic acid +acetyl glucosamine) is the mucopolysaccharide met in extracellular fluid of animal tissues, vitreous humor of eye, synovial fluid, cerebrospinal fluid, etc. It also occurs in cementing material between animal cells as well as inside cell coat.
(8) Husk of Plantago ovata contains mucilage which is used medicinally in treating intestinal problems. It relieves irritation. Mucilage present in Aloe barbadensis reduces inflammation. It is also purgative.
(9) Mucopolysaccharide heparin (α-1, 4 glucosamine + glucuronic acid) is blood anticoagulant.
(10) Agar. Marine brown and red algae, called sea weeds, yield mucopolysaccharides of commercial value, e.g., agar, alginic acid, carragenin, etc.
Agar (agar-agar) is polymer of D-galactose, 3-6 anhydro L-galactose having sulphate esterification after every tenth galactose unit. It is used as culture medium in the laboratory, as laxative, stiffening, stabilising and emulsifying agent. It is obtained from cell wall of some red alae like Gracilaria, Gelidium and Gelidiella.
Pectin (Pectic Compounds):
It is an acidic polysaccharide that occurs in the matrix of cell wall and middle lamella (as calcium pectate). Pectin is soluble in water and can undergo ↔ sol gel interchange. Pectin is formed of galacturonic acid, galactose, methylated galacturonic acid and arabinose. It is used in making Jelly and Jams.
Hemicellulose:
It is a mixture of polysaccharides of xylans, mannans, galactans, arabino-galactans and glucomannans. Hemicellulose occurs in the cell wall where it forms a link between pectic compounds and cellulose micro fibrils.
Peptidoglycan (Murein, Mucopeptide):
It is formed of heteropolysaccharide chains cross-linked by short peptides (generally tetra peptides). Heteropolysaccharide chains are formed of two alternate amino-sugar molecules, N-acetyl glucosamine (NAG) and N-acetyl muramic acid (NAM).
Lipopolysaccharide:
It is a complex formed of lipid and polysaccharide which forms the outer membrane of Gram -ve bacteria. There is a glycolipid responsible for endotoxic activity, a core polysaccharide and an antigen specific variable chain. Lipopolysaccharide induces fever, shock and other toxic effects.
Mucoproteins (glycoproteins):
The protein with conjugated monosaccharide’s form mucus. These are found in stomach, nasal secretion, intestine, vagina and are antibacterial and protective in function.
